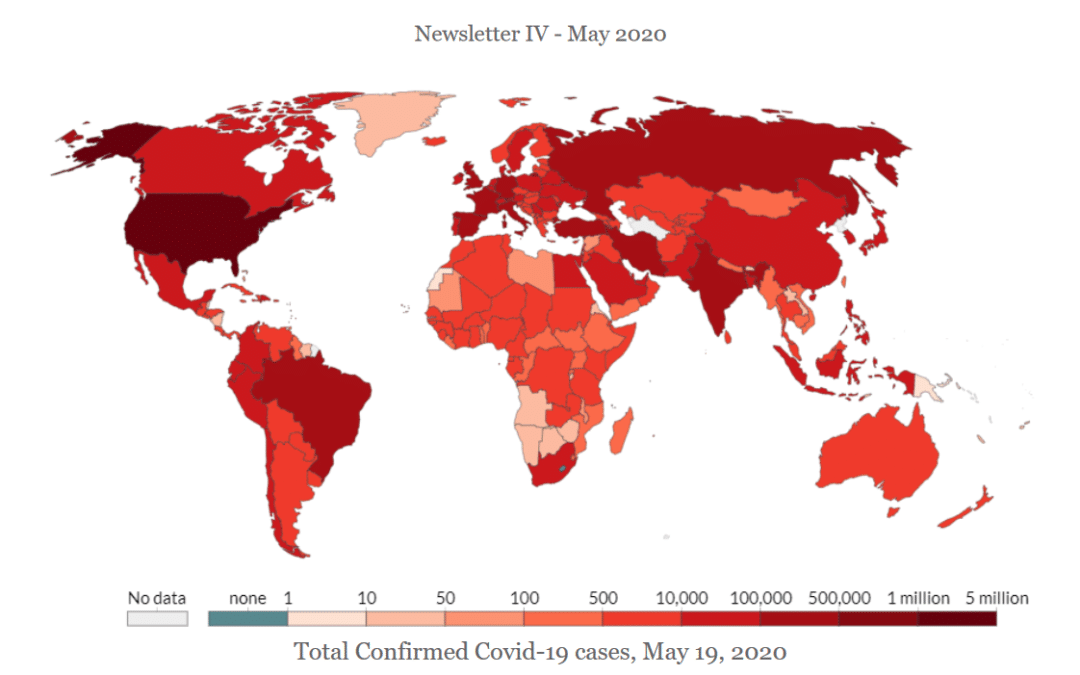
by Deb Dulal Das | May 30, 2020 | Blog
Ventilators are medical devices to help critical patients breathe in and breathe out. During COVID-19 pandemic, 1 out of 6 patients gets ventilators. However, owing to the rising numbers, hospitals are suffering from a dearth of ventilators even though the USA and...


